
Abstract
The project is devoted to design, synthesis and structural characterization of metal-organic frameworks (MOFs) with controlled hydrophobicity required for certain applications such as gas storage, drug delivery systems, self-compatibilizing fillers for special energy composites, supercapacitors, etc. Different from the approaches reported in literature consisting in attaching hydrophobic groups near coordination sites, or post-synthetic grafting of such groups onto linkers, here will be used mainly ligands with siloxane spacers having attached to the silicon atoms one of the highest hydrophobic group, methyl, but also some derivatives inserting more longer (octyl), more rigid (phenyl, diphenyl), more polar (chloropropyl) or more hydrophobe (trifluoropropyl) groups in order to fine tune moisture stability of the resulted MOFs but also their lipophilicity and crystallinity. The high flexibility of the siloxane backbone allows the organic groups to be arranged and presented to their best effect. In addition, metals in high oxidation state will be used. The key steps in achieving the project objectives consist in engineering the spacer by using new approaches in silicones chemistry (i.e., Piers-Rubinsztajn reaction), attaching coordination groups (by thiol-ene addition or nucleophilic substitution), construction of MOF's and their isolation in a form accessible to characterize accurately. Thus original polydentate ligands mainly consisting in polycarboxylic acids and N-donor heterocycles with controlled diorganosiloxane or silane spacers will be obtained and used to built MOFs. The rare examples of assembling using the flexible linker, apart from those published by the authors of this proposal, and limited investigation in the field opens the innovative perspective for new knowledge and unique properties of MOFs.
Stage I (2017): Synthesis of poly carboxylate ligands siloxane spacers
Deadline: 31.12.2017
Expected Results:
• Six siloxane ligands with carboxyl groups•At least one published article associated with the project
•At least one article sent to the publication and associated with the project
Expected Research Activities
1.1.Functionalization of siloxane substrates with carboxyl groups by click chemistry (thiol-ene addition): thiol-ene addition of 3-mercaptopropionic acid and thioglycolic acid to the vinyl groups of the siloxane substrates with different architectures: cyclic (V3, V4) or linear (DVS); characterization of the resulted ligands by elemental, spectral (IR, NMR, UV), and X-ray (if applicable); evaluation of hydrophilic-hydrophobic balance of the obtained ligands on the basis of established structure; study of behavior in solution of the obtained ligands.1.2.Carrying out tests for the complexation of metal ions with polycarboxylic silicone ligands; characterization of complexes obtained by elemental analysis, spectral (IR, NMR, UV) and X-ray single crystal diffraction (where applicable).
1.3.Reporting and dissemination activities: centralization of data, interpretation and preparation of their reporting, publication and presentation at scientific.
Stage II (2018): Preparing of N-heterocycle siloxane ligands
Deadline: 31.12.2018
Expected Results:
● At least four new ligands● At least five presentations at scientific events
● At least three published articles to be associated with the project
Expected Research Activities
Act 2.1 - Spacers: synthesis of bis(vinyl)siloxanes with different spacers (e.g., methylphenyl, diphenyl siloxanes); characterization of proligands obtained by spectral analysis (IR, NMR), GPC.Act 2.2 - Functionalization of siloxane substrates with N-heterocycles by "click chemistry" (thiol-ene addition); characterization of resulting ligands by elemental and spectral (IR, MN, UV-Vis) analysis and single crystal X-ray diffraction (if applicable); evaluation of hydrophilic-hydrophobic balance of ligands obtained on the basis of the established structure; the study of the behavior of ligands obtained in solution.
Act 2.3 - Synthesis of N-heterocyclic ligands by nucleophilic substitution of chloroderivatives with thiol derivatives; optimizing the synthesis; characterization of reaction products by elemental and spectral (IR, NMR) analysis and single crystal X-ray diffraction (if applicable).
Act 2.4 - Reporting and dissemination activities: Centralizing data, interpreting and preparing them for reporting, presenting to scientific events, and publishing.
Stage III (2019): Synthesis of metal complexes and metal-organic networks of MOF type
Deadline: 31.12.2019
Expected Results:
● At least five new metal complexes or MOFs● At least five presentations at scientific events
● At least two published scientific articles to be associated with the project
Expected Research Activities
Act 3.1 - Synthesis of metal complexes and metal-organic networks (MOFs): establishment of the experimental program; realization of syntheses according to experimental program; purification and structural characterization of reaction products by appropriate techniques; verifying reproducibility of syntheses and products can be found to be optimal.Act 3.2 - MOFs evaluation: assessment of behavior in wet environment through water vapor sorption experiments combined with IR spectroscopy; estimation of porosity and BET surface for the compounds obtained; analyzing the results and identifying the conditions that allow obtaining the compounds with the highest porosity and the specific surface and the lowest water retention capacity; tests to identify applicative potential.
Act 3.3 - Reporting and dissemination activities: data centralization, interpretation and preparation for reporting, presentation at scientific events and publication
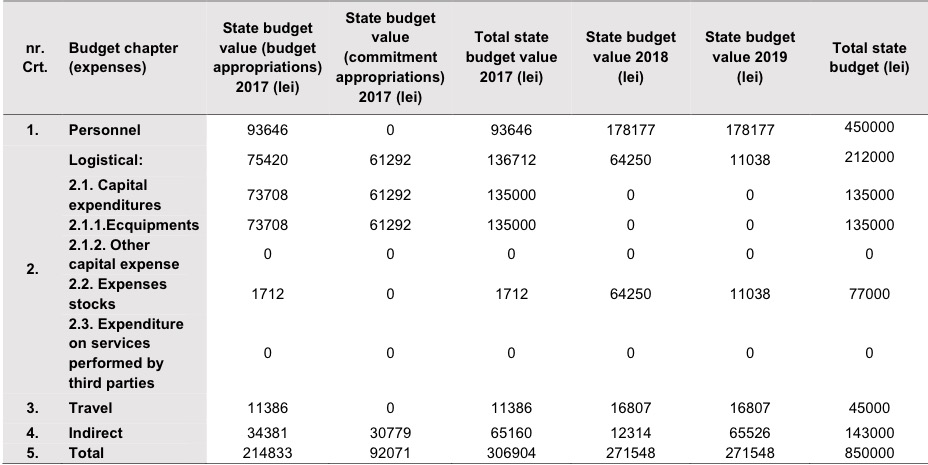
Budget
 Download table in pdf
Download table in pdf